 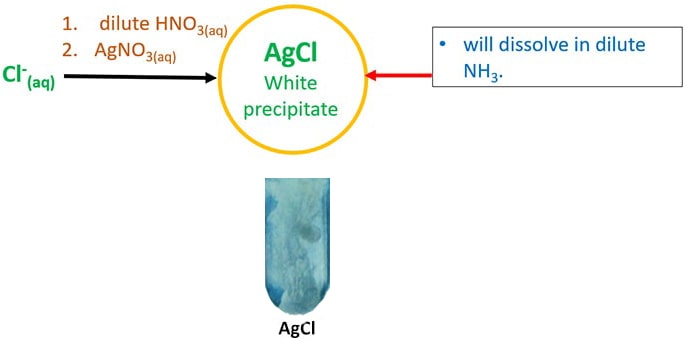
The order of the color intensity in the covalent compound is shown below. Thus, the silver salts like AgBr and AgI are colored. With the increase in polarization, the intensity of compound color increases. The property of anion to get polarized by the deformation of the electron cloud formed by theĬation is known as polarization. With the increase in the size of anions, the polarization of anions increases. On moving down in the group the atomic size of the increases, the order is given as shown below. As no unpaired electrons are present d-d transition cannot take place.The color of the compound AgCl is white but silver salt of AgBr and AgI are colored. The electronic configuration of silver is $4$, all the electrons are paired. Fluorine is the smallest halogen ion and iodine is the largest halogen ion. In the given compounds, halogens ions are present. Note: all nitrates, sodium, potassium and ammonium containing ionic compounds are aqueous.Hint: As we know that on moving down in the group, the size of the anions increases. HSC Solubility Table Between Cations and Anions Temperature increase due to neutralisation White precipitate in alkaline conditions. If no precipitate forms, acetate ions are present.Chloride ions are identified through addition of AgNO 3.Phosphate ions are identified through addition of alkaline Ba(NO 3) 2.Sulfate ions are identified through addition of Ba(NO 3) 2.This test does not work if phosphate ions are also present. Hydroxide ions are identified by adding Cu 2+ ions to produce a blue precipitate.Carbonate ions are identified through neutralisation with a strong acid (HNO 3) to produce CO 2.Thus, salts containing phosphate are basic HNO 3 is used instead of HCl & H 2SO 4 because the latter two introduce precipitable anions into the unknown solution which may produce a false positive result.Adding nitric acid to produce carbon dioxide, causing bubbles to form.Red litmus paper turns blue in basic solution Production of acetic acid at high concentrations produces a distinct vinegar smell. Adding nitric acid shifts acetate ion’s ionisation equilibrium to the right (Le Chatelier’s principle) to form more acetic acid.When dissolved in water, it exists in the following equilibrium: Indicator test – acetate is the conjugate base of acetic acid (weak acid).Precipitation tests are not effective for identifying acetate ions as salts containing acetate are usually quite soluble.Indicator test – hydroxide ions cause solution to become alkaline (pH > 7).Precipitation test – hydroxide ions produce soluble ionic compounds with most cations except:.Pb(NO 3) 2 will produce a bright yellow precipitate (PbI 2) AgNO 3 will produce a yellowish precipitate (AgI). AgNO 3 will produce a cream-coloured precipitate (AgBr). Precipitation test – adding silver ions e.g.sunlight) will cause it turn grey as AgCl decomposes into silver metal. Exposing this precipitate to UV light (e.g. AgNO 3 will produce a white precipitate (AgCl). This video explores qualitative investigations that can used to test for the presence of anions including: 
Testing for Anions Using Precipitation and pH Indicators anions: chloride (Cl–), bromide (Br–), iodide (I–), hydroxide (OH–), acetate (CH3COO–), carbonate (CO32–), sulfate (SO42–), phosphate (PO43–).HSC Chemistry Syllabus Conduct qualitative investigations – using flame tests, precipitation and complexation reactions as appropriate – to test for the presence in aqueous solution of the following This is part of the HSC Chemistry course under Module 8 Section 1: Analysis of Inorganic Compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
